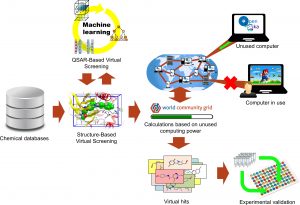
Em maio de 2016, a Universidade Federal de Goiás (UFG), em parceria com a World Community Grid (WCG), da International Business Machines (IBM), iniciou o projeto OpenZika, que visa identificar substâncias com potencial para tratar pessoas infectadas pelo vírus Zika. De uma lista inicial de aproximadamente 7.600 compostos, dentre eles fármacos já aprovados para uso em humanos, cinco foram selecionados e estão em fases de testes in vitro na University of California San Diego (UCSD). Agora, o grupo acaba de descobrir mais nove substâncias potenciais que serão testadas.
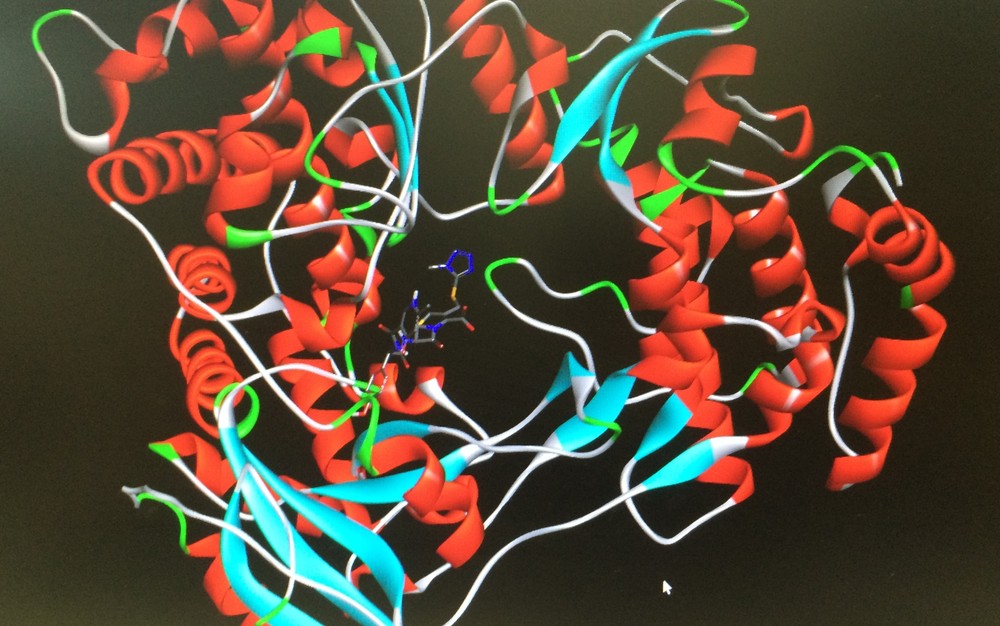
A descoberta surgiu em uma segunda leva de pesquisas com 260 compostos adicionais que foram testados por meio de uma triagem virtual, maior e mais diversa, contra as estruturas cristalinas da proteína helicase NS3 do vírus Zika, ligadas ao ácido ribonucleico (RNA). Os compostos também serão encaminhados para a universidade californiana ainda neste mês de março, na busca do desenvolvimento de um medicamento antiviral.

Leia mais